DKFZ Heidelberg - BioQuant Heidelberg - ZMBH Heidelberg - UKE Hamburg - CSCC Jena
e:Med Systems Medicine Program
CancerTelSys - Identifying cancer telomere maintenance networks for diagnosis, prognosis, patient stratification and therapy response prediction
Research mission
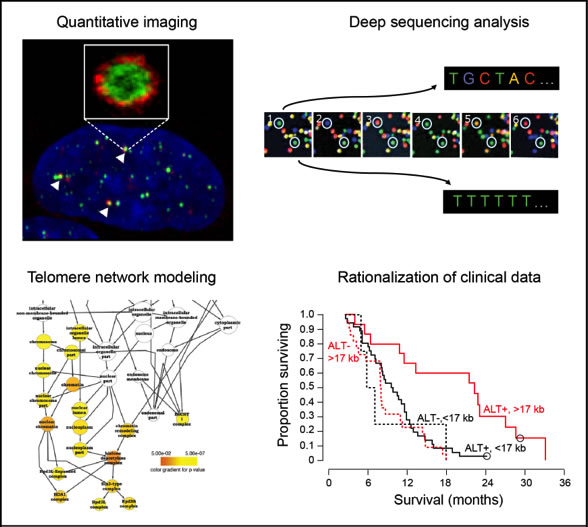
Cancer cells require a telomere maintenance mechanism (TMM) to avoid cellular senescence and apoptosis induced by the replicative shortening of their chromosome ends. Frequently, telomerase is reactivated to extend the telomeres. However, in 5-25 % of the cases (depending on tumor entity) alternative lengthening of telomeres (ALT) pathways exist that operate via DNA repair and recom- bination processes. The identification of the active TMM provides highly valuable information from which prognostic and stratifying analysis schemes can be derived that address the clinically ob- served tumor heterogeneity. For example, the presence of a specific TMM in glioblastoma and lymphoma is a prognostic marker for patient survival. Since cancer therapies targeting telomerase can select for the emergence of an ALT-positive cancer cell population, specific assays and treat- ments for the different TMM subgroups are needed to diagnose and target these recurrent tumors. In CancerTelSys, we will implement a systems medicine approach that integrates a bioinformatical analysis of (epi)genomic data with automated high-throughput quantification of 3D fluorescence microscopy images to develop network models that describe the active TMM of a given tumor patient sample. In an iterative cycle between experimental studies and modeling work, we will derive a TMM classification scheme for the TMM status in glioblastoma and prostate cancer samples for which we have already identified the presence of clinically relevant subgroups with different TMMs. The TMM classification will be applied in a clinical setting for prognostic, predictive and stratifying analysis of tumor samples. Furthermore, we will develop strategies to combine existing telomerase inhibitors with novel approaches that target ALT and promote telomere repeat resection for improv- ing the application of TMM inhibition in cancer therapy and will identify specific drug targets for the different TMM subgroups based on the network models.